Structure dot oxygen electron write o2 lewis draw gas diatomic dioxygen symbol electrons sodium pc O2 oxygen molecule electrons geometry diatomic techiescientist hybridization valence covalent bonding O2 lewis structure: how to draw the lewis structure for oxygen gas
Oxygen, O2, molecule model and chemical formula. Also dioxygen
Oxygen electron o2 electrons valence molecular structures oxide molecule calcium hybridization socratic compound atomic configuration techiescientist Oxygen periodic valence electrons Oxygen dot diagram wiring diagrams lewis electron dot
O2 lewis structure, molecular geometry, and hybridization
What is the electron dot diagram for an oxygen atom?O2 electron molecule oxygen valence chemistry Dot o2 oxygen diatomic electron bonds carbon electrons nitrogen learnwithdrscottLewis dot structure – easy hard science.
Lewis dot diagram for oxygenLewis dot structure for oxygen atom (o) O2 oxygen molecule molecular structure model formula gas chemical stick ball diatomic air 3d stock dioxygen also geometric alamy highlyOxygen, o2, molecule model and chemical formula. also dioxygen.

Oxygen model drawing bohr lewis dot structure atom draw electrons show difluoride valence many
Dot oxygen electron wiringElectron fluorine oxygen configuration configurations chemistry atomic neon number gas electronic atom electrons arrangement shell orbital elements atoms full valence Orbital molecular diagram mo oxygen o3 o2 configuration electron orbitals bonding diagrams draw energy dioxygen level edu via uwimona jmMakethebrainhappy: the lewis dot structure for o2.
Chemistry: september 2014 .
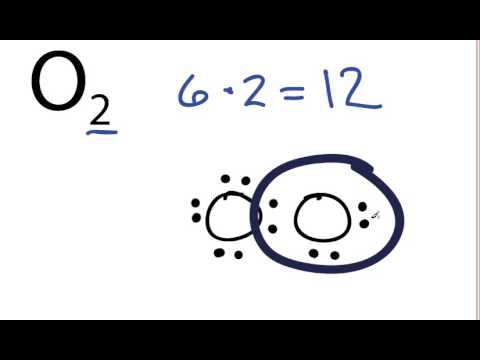

Lewis Dot Diagram For Oxygen - Wiring Site Resource

Lewis Dot Structure – Easy Hard Science

Oxygen Dot Diagram Wiring Diagrams Lewis Electron Dot - Electron Dot

Chemistry: September 2014
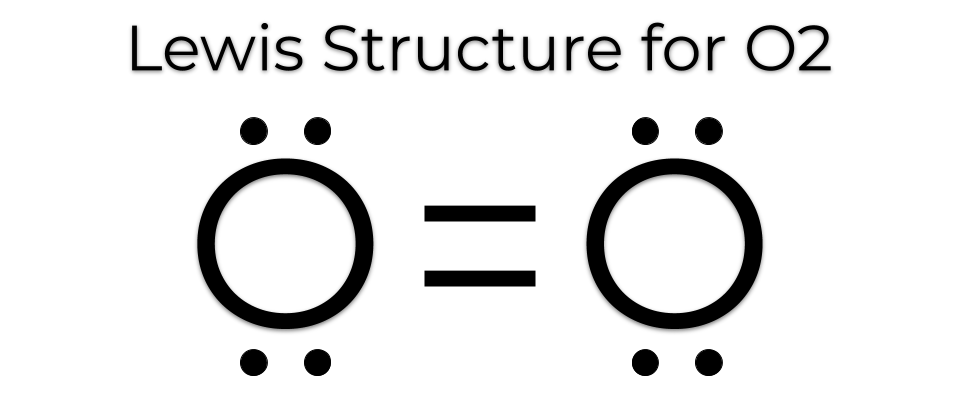
MakeTheBrainHappy: The Lewis Dot Structure for O2

Oxygen - Chemwiki
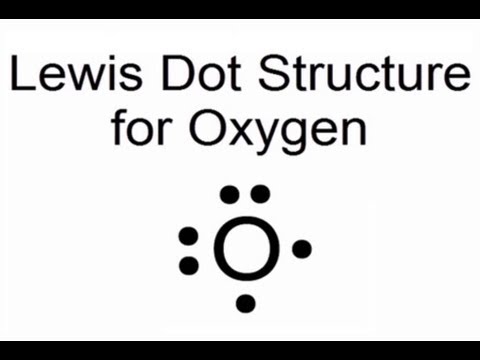
Lewis Dot Structure for Oxygen Atom (O) - YouTube

O2 Lewis Structure, Molecular Geometry, and Hybridization - Techiescientist

What is the electron dot diagram for an oxygen atom? | Socratic